- Home
- Services
- About
- News
- Contact
- Intel hd graphics 4000 driver updates
- Avs video editor 8-0 activation code
- How to use vocaloid 4
- Capture one pro trial
- Download vray sketchup 2016 32 bit with keygen
- Justin bieber never let you go 2015
- 60 inch tv
- Philips oneblade
- Reason core security keep or uninstall
- Myocardial perfusion imaging
- Pokemon x and y anime brock
- How to install games on a usb advance ps2
- Paddle agitator design
- Apple tv

All four DMDPA derivatives exhibited significantly lower heart/liver radioactivity uptake ratios (0.6, 0.4, 0.7 and 0.6, respectively) compared to that of DMDPA (1.2). The other two were labeled using carbon-11: methyl-(3-fluorophenyl)-methylphenylammonium trifluoromethanesulfonate (3-F-DMDPA) and methyl-(4-fluorophenyl)-methylphenylammonium trifluoromethanesulfonate (4-F-DMDPA). Four fluorinated DMDPA derivatives were synthesized, two were labeled with fluorine-18: fluoroethyl-methyldiphenylammonium trifluoromethanesulfonate (FEMDPA) and fluorobuthyl-methyldiphenylammonium trifluoromethanesulfonate (FBMDPA). Organ uptake after injection of fluoroethylquinolinium acetate (FEtQ) was examined ex vivo. The dynamic distribution in vivo, following injection of each derivative into male SD rats, was evaluated using small-animal dedicated PET/CT.
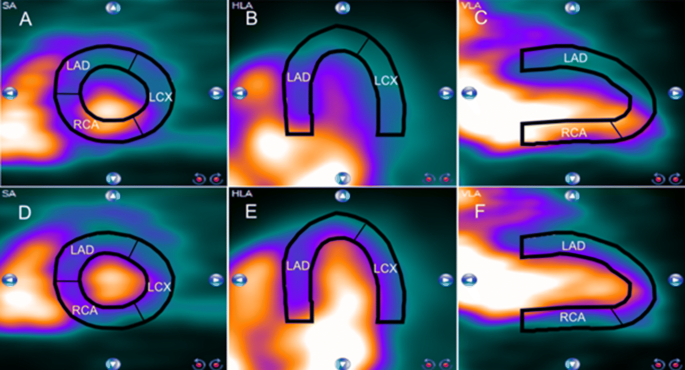
Four fluorinated DMDPA derivatives and two quinolinium salt analogs were radiolabeled. The current study aimed to increase the clinical applicability of PET-MPI by designing and synthesizing fluorinated ammonium salt derivatives. We previously presented the radiolabeled ammonium salt -dimethyl diphenylammonium trifluoromethanesulfonate (DMDPA) as a potential novel PET-MPI agent.
- Home
- Services
- About
- News
- Contact
- Intel hd graphics 4000 driver updates
- Avs video editor 8-0 activation code
- How to use vocaloid 4
- Capture one pro trial
- Download vray sketchup 2016 32 bit with keygen
- Justin bieber never let you go 2015
- 60 inch tv
- Philips oneblade
- Reason core security keep or uninstall
- Myocardial perfusion imaging
- Pokemon x and y anime brock
- How to install games on a usb advance ps2
- Paddle agitator design
- Apple tv
